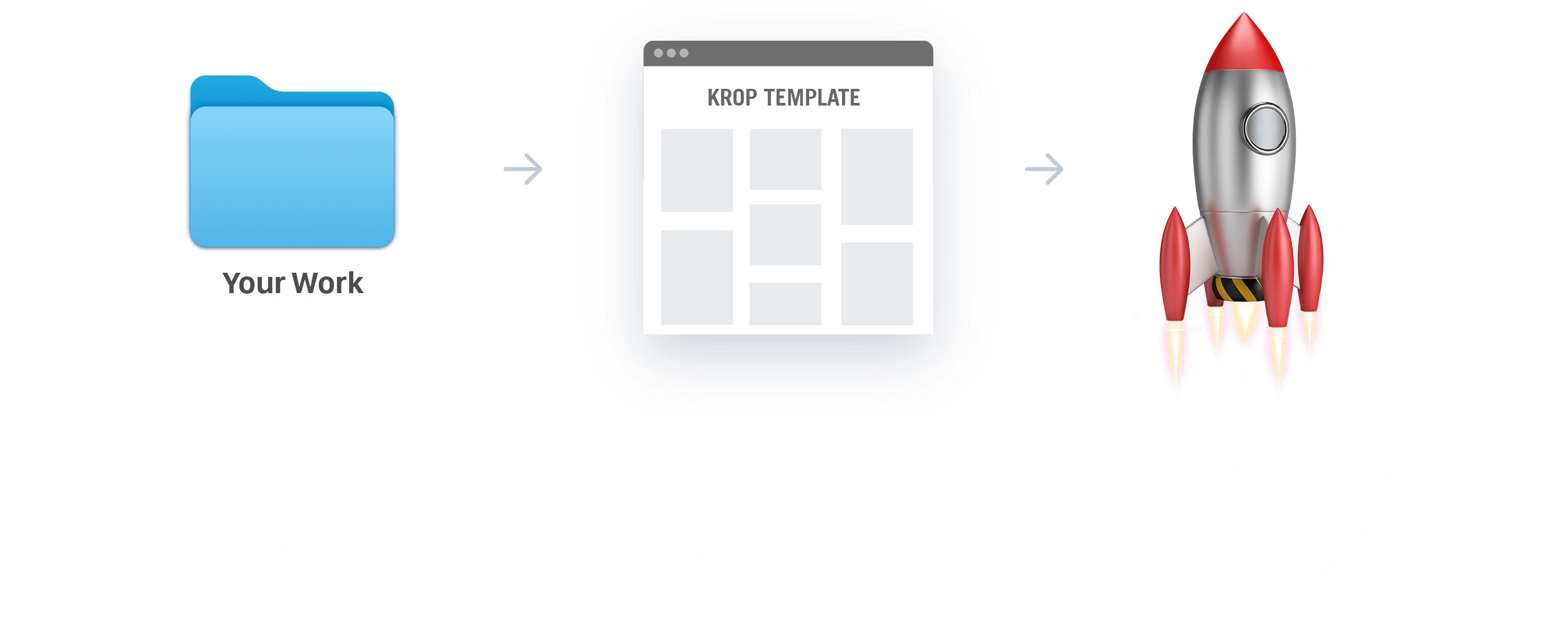
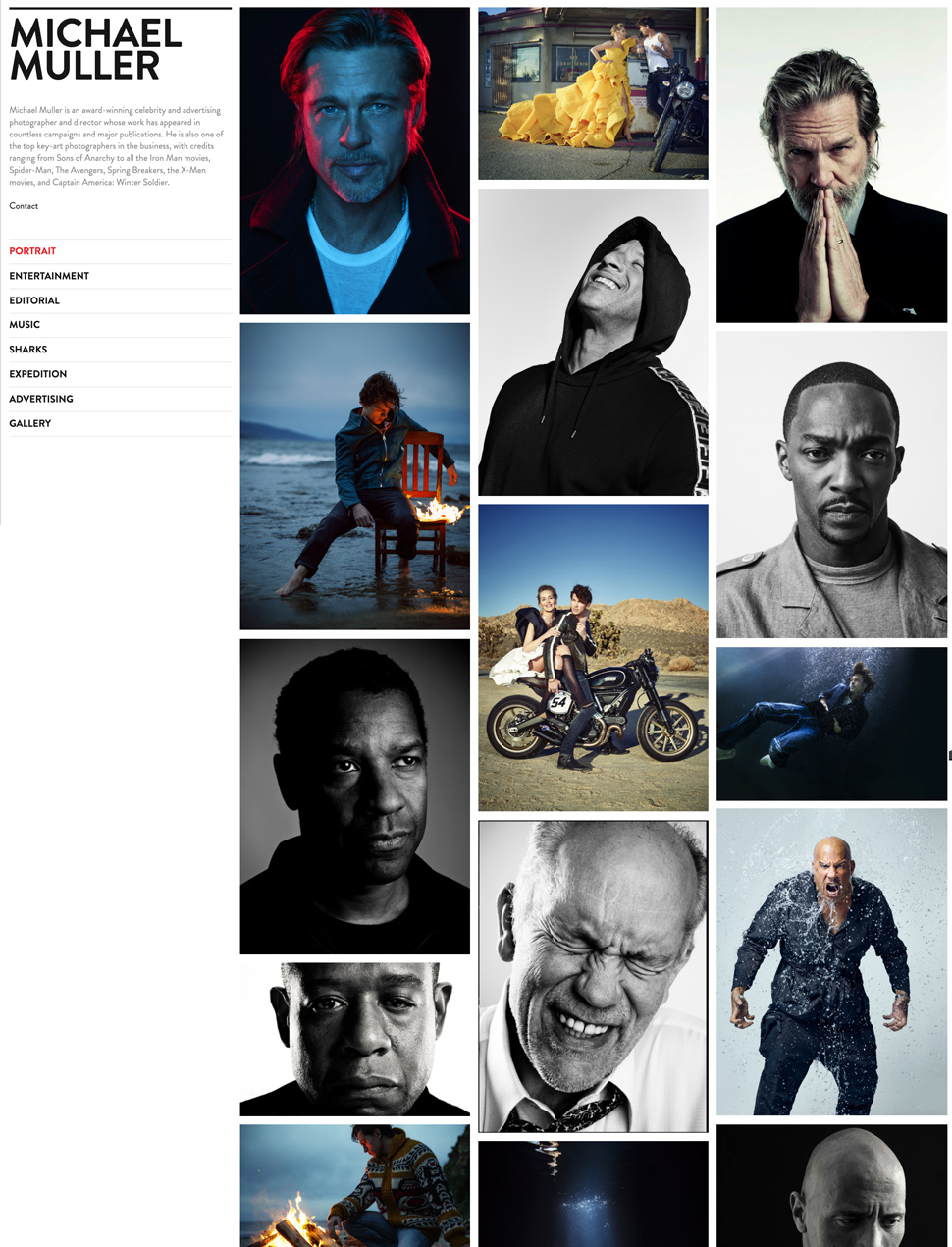
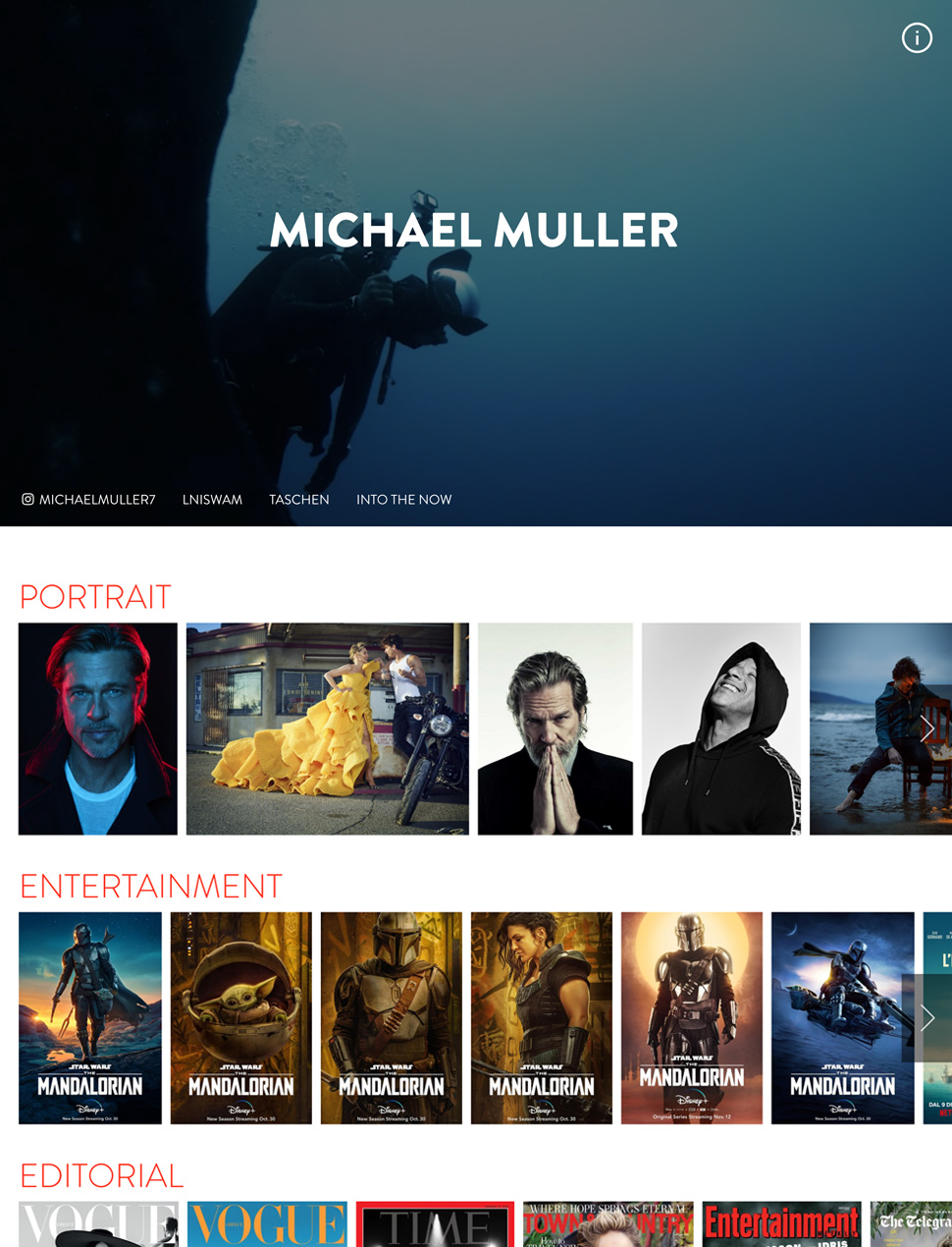
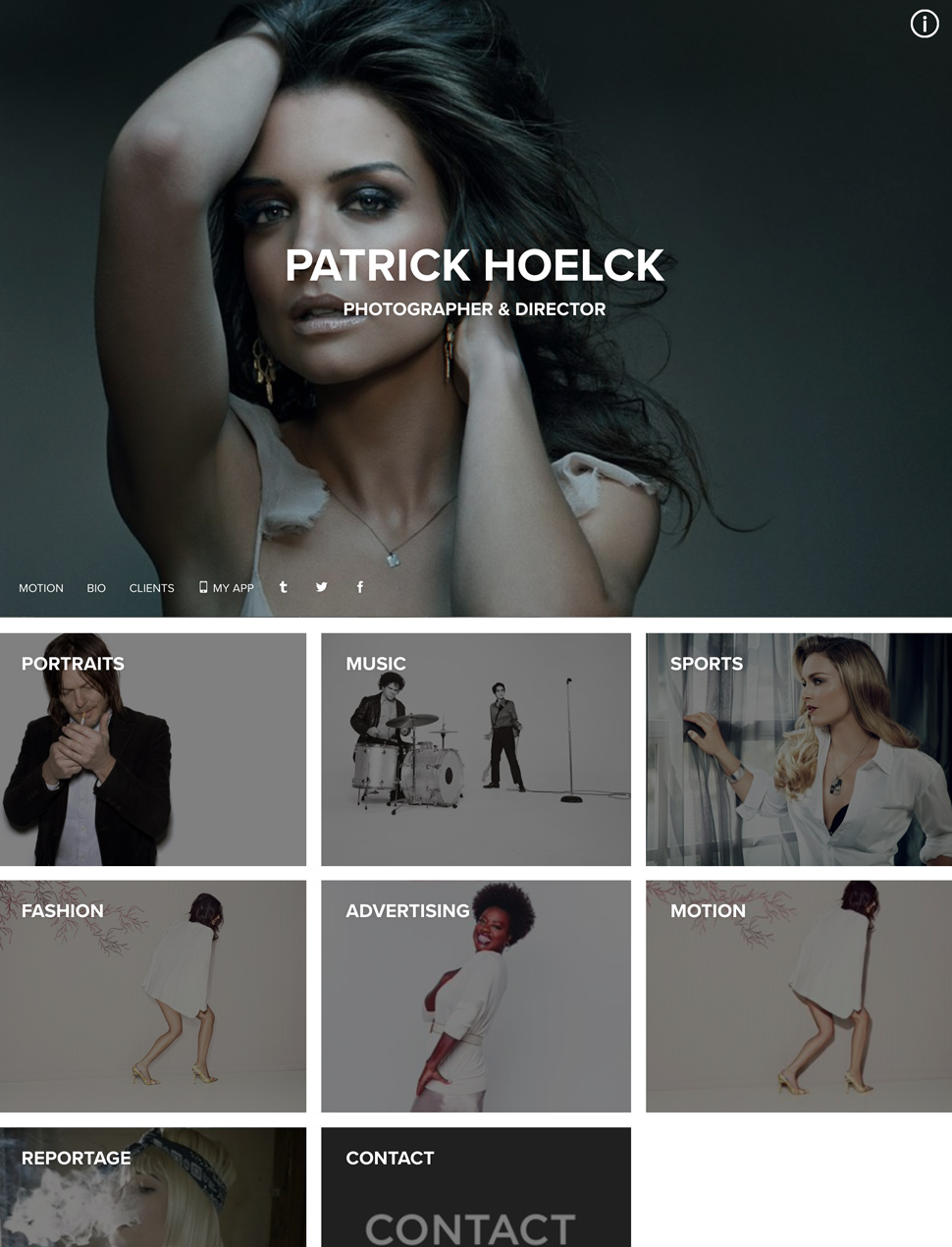
The Easiest, Most Beautiful Portfolio Website Builder
Krop takes your creative work and magically turns it into a stunning portfolio website.
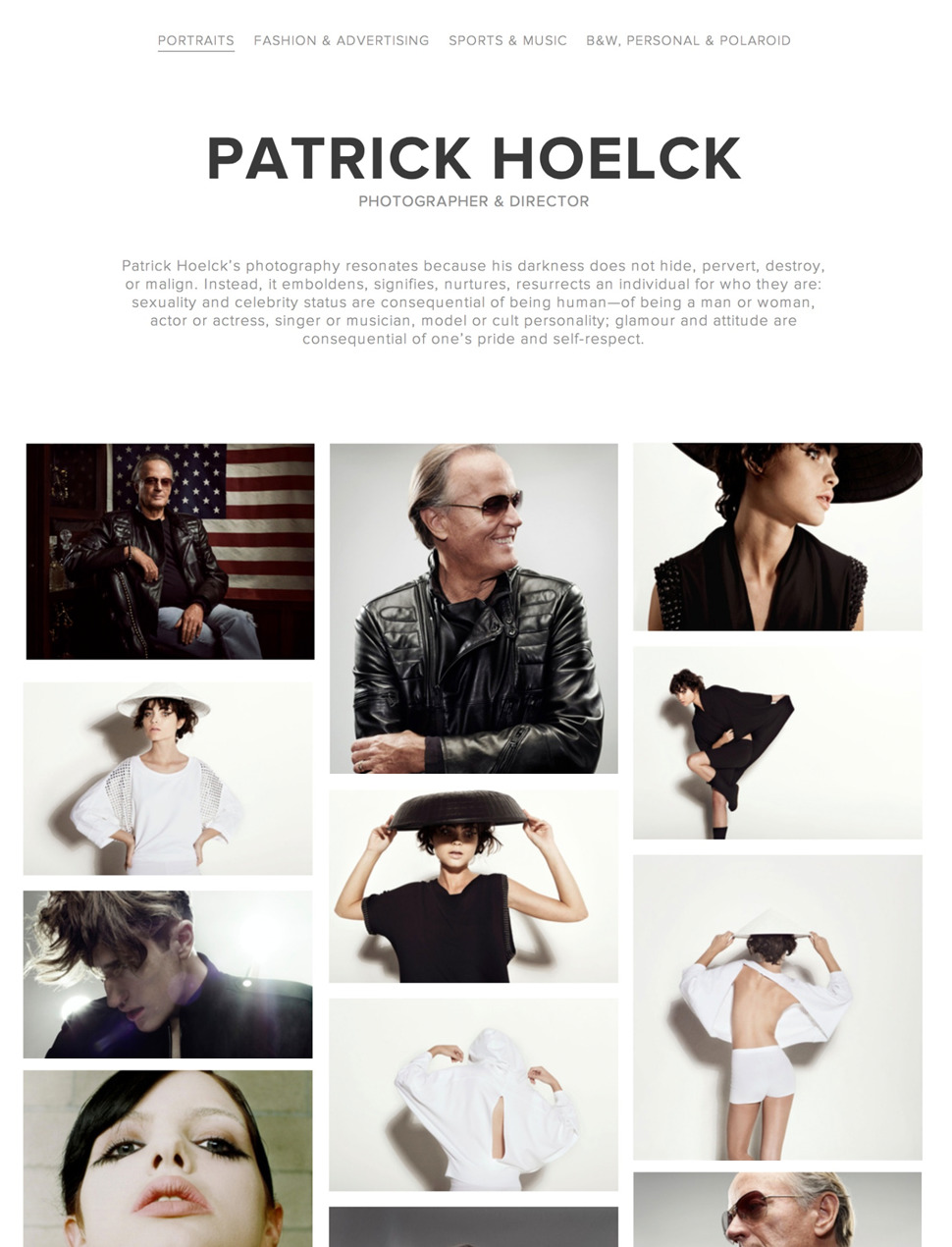
Made for Visual Thinkers
Photographers, designers, illustrators, and other creative professionals use Krop to build and manage their own portfolio websites without having to learn a complicated platform or needing to hire a web developer.
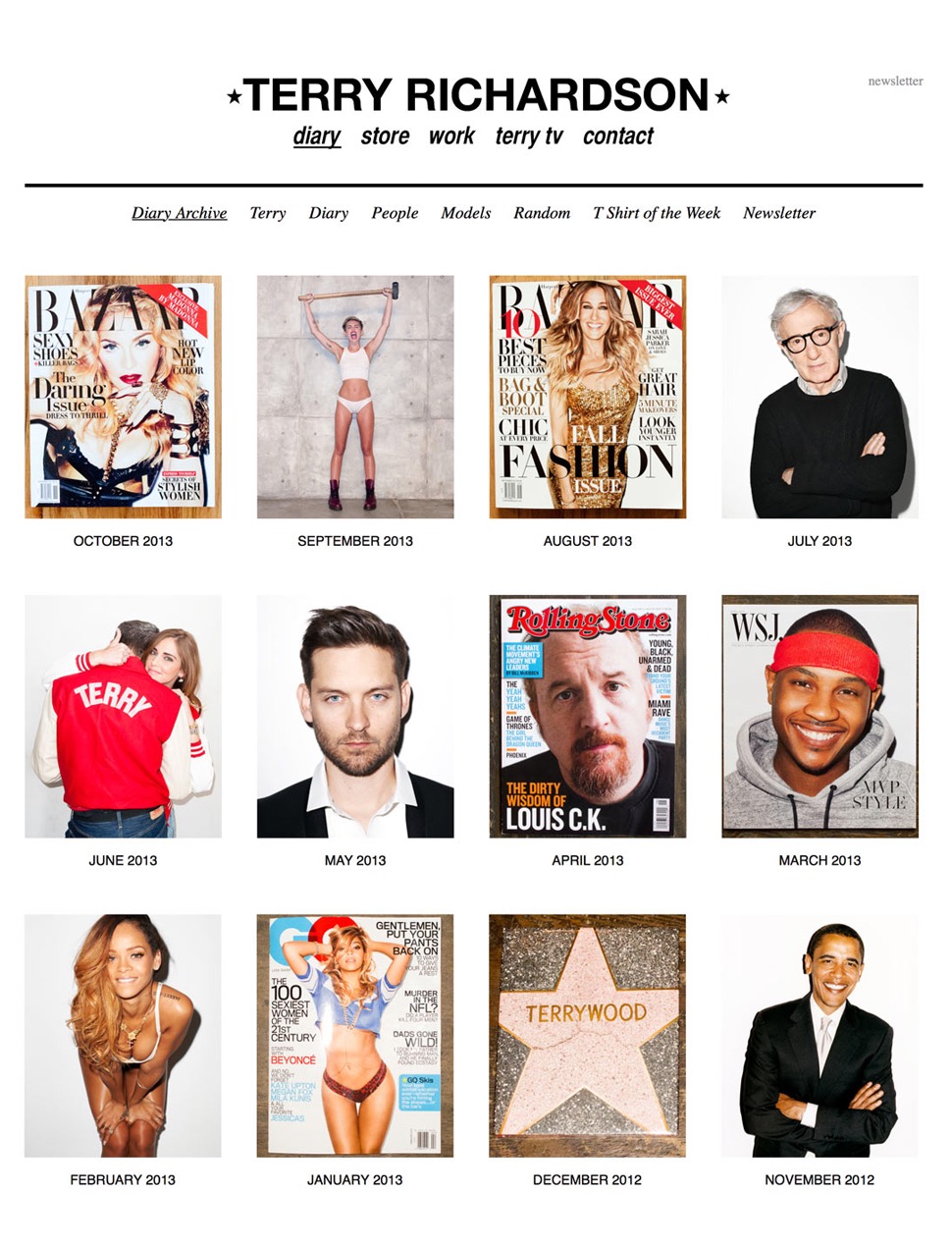
Organize Your Work
Curate your creative work as easily as sorting files into folders.
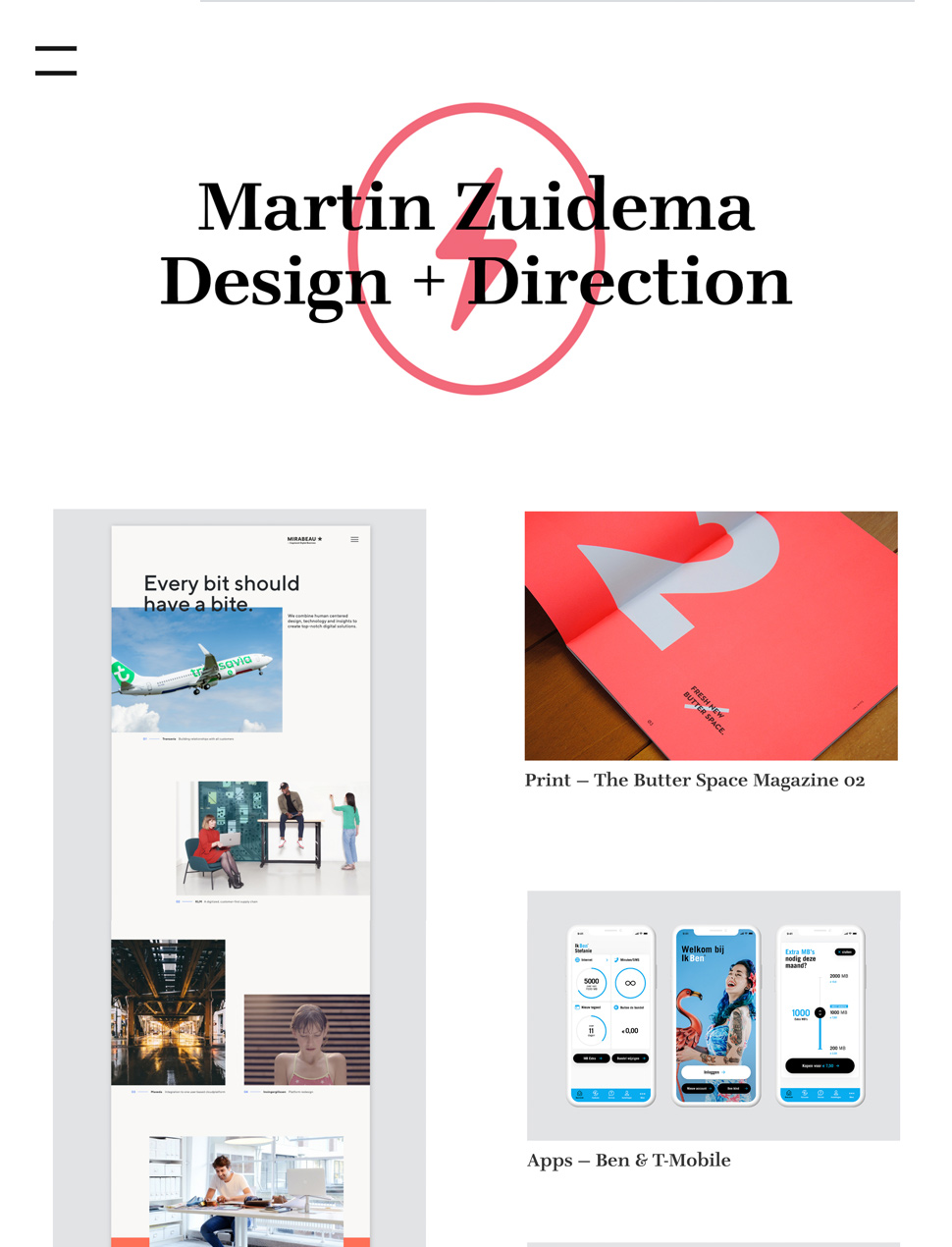
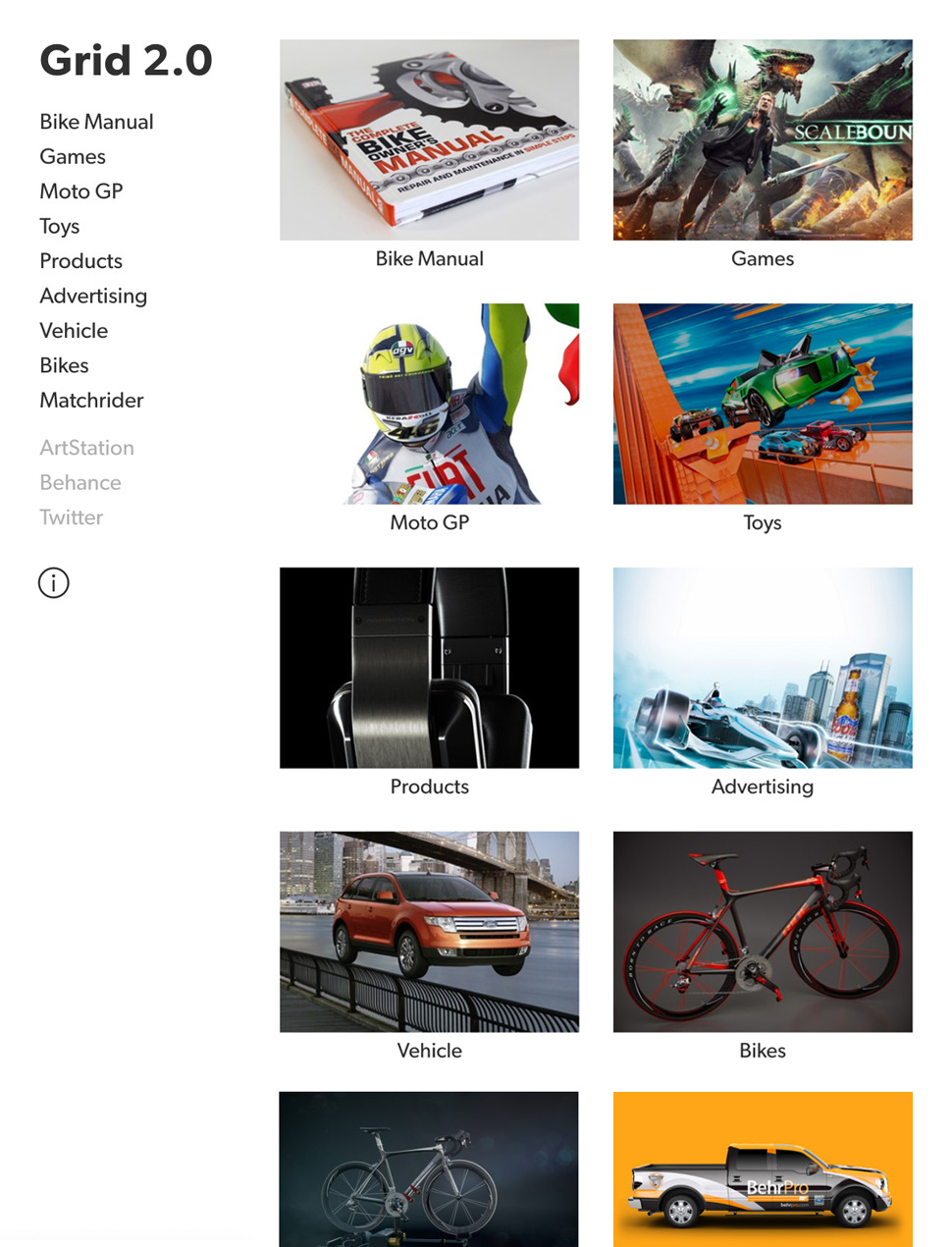
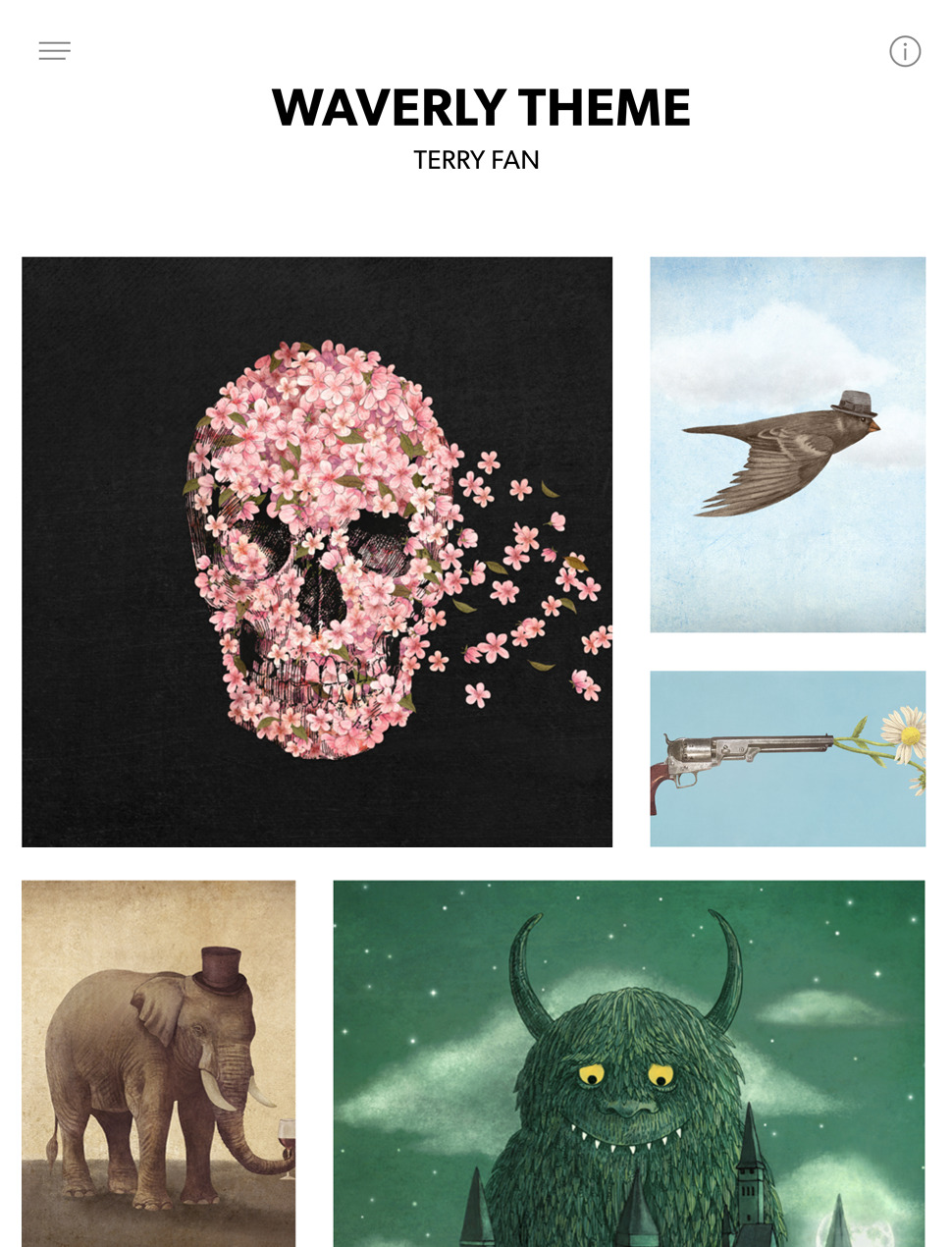
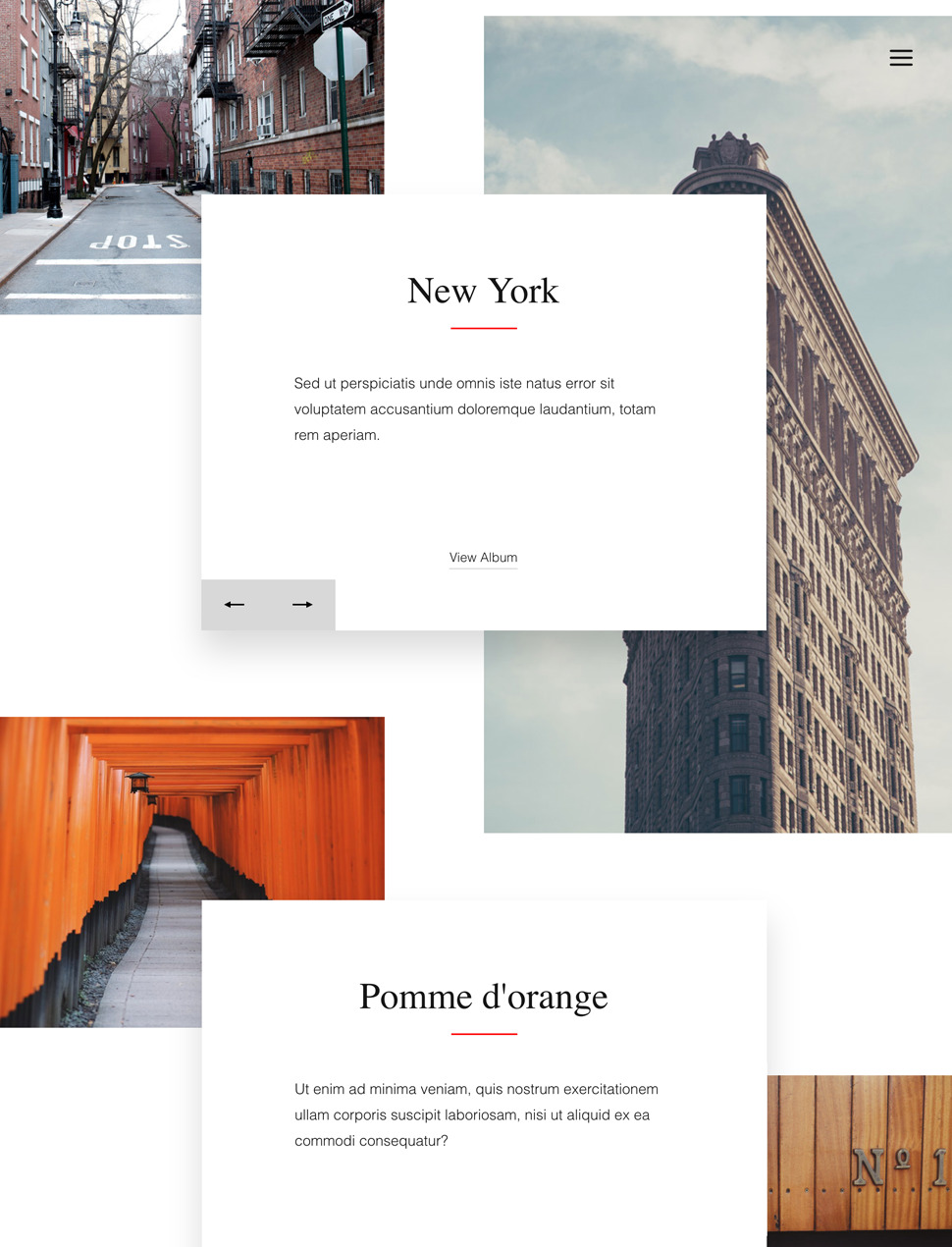
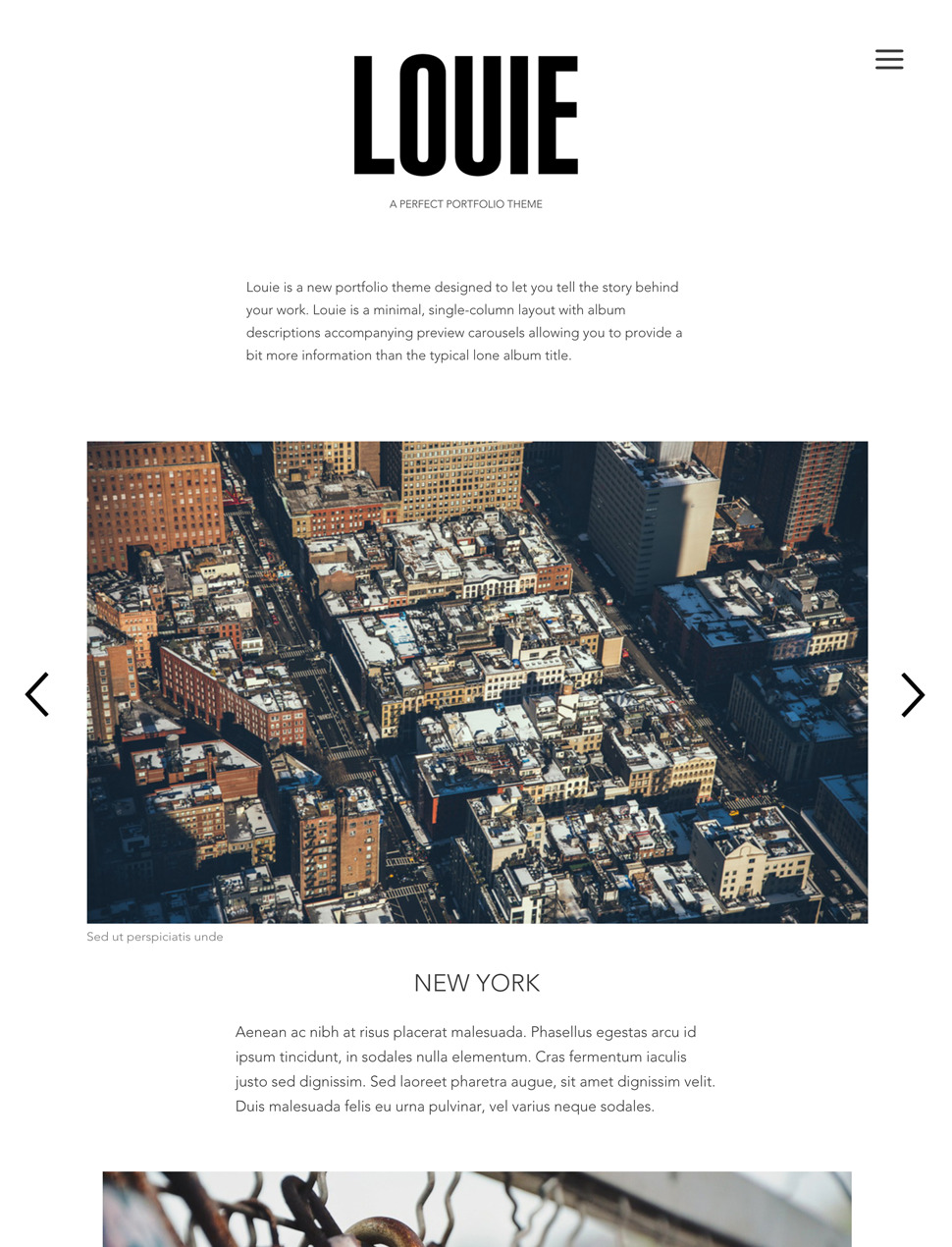
Designer Templates
What you see is what you get. Beautiful templates that you can try with a single click.
Unlimited Images
Never worry about running out of storage space or having to upgrade plans.
Instagram Integration
Automatically display your latest Instagram posts, right on your portfolio.
No Code, DIY For Real
No HTML or CSS knowledge necessary. Krop simply uses your albums to generate an awesome website.
Powerful Customizations
Style your template with a logo, fonts, and colors using our easy editor.
Private Albums
Password protect individual albums or your entire portfolio to share with clients privately.
Video Backgrounds
Add life to your website with a custom video background, or select one from our gallery.
Free Domain Name
Register www.yourname.com free for the first year.
Simple Pricing
Free Trial, then $8.25/month
Discount applied for annual billing. Cancel anytime.
Drag, Drop, Launch.
Our most important feature is simplicity. Our platform is for creatives, not developers. We want you to love your portfolio, launch it, and get more jobs!